
The picture of electrons revolving round a nucleus on select avenues has become the icon of the atomic age. This conclusion led to the theory that electrons exist in energy levels around the positive nucleus and have their own distinct properties in each of their energy levels. The model of Niels Bohr (18851962) for the atom is since long just the one and only conception for atoms of the vast majority of educated people. This theory was adopted by Niels Bohr in 1913 who theorised that electrons could orbit the nucleus in a circular orbits and that the distance of the electron to the nucleus was fixed unless it moved between energy levels with the absorption or emission of light. Max Planck and Albert Einstein in the field of physics postulated that light energy can be absorbed and emitted as quanta. 
It was not until the earlier 20th Century that the scientific community arrived at the modern day atomic model. Das Atommodell von BOHR, so wie es oft genau wie in der Animation in Abb. 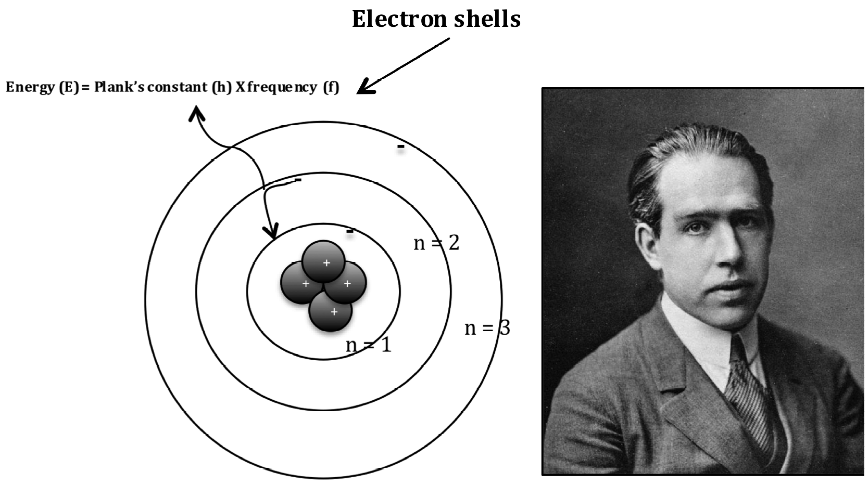
Now the atomic model had a central particle and electrons around it, reversing he plum pudding model of Thomson. 1 Atommodell von BOHR mit den auf diskreten Bahnen um den positiv geladenen Kern kreisenden Elektronen. He named this new fundamental particle as a proton. Rutherford conducted a number of experiments with hydrogen nuclei and nitrogen in air using alpha particles and after a number of theories concluded that the hydrogen atom made up other atoms. Rutherford further followed this up in 1917 when he proved that a hydrogen nucleus (1 proton) is present in other nuclei of different elements most notably nitrogen gas in the air.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
